When she enrolled at UT with a love of genetics, Hannah Boyd thought she was destined for medical school. However, she quickly began to feel that her pre-med coursework was missing something.
“I realized that biology didn’t have enough math and physics for me,” Boyd said. “I kind of wanted to do something that felt more applied and problem solving-based.”
She switched tracks, becoming a chemical engineering major with a concentration in biomolecular engineering, a field with a more satisfying blend of math and biology.
Now a junior, Boyd is working to improve treatments for antibiotic-resistant bacterial infections in the laboratory of Cong T. Trinh, a Ferguson Faculty Fellow and professor in the Department of Chemical and Biomolecular Engineering.
“Undergraduates are often not given the opportunity to discover what they are capable of,” said Trinh. “Engaging undergraduates in lab research is the best way to fill that gap. Curiosity and creativity lead to innovation.”
CRISPR-Cas Kills Antibiotic-Resistant Bacteria
The bacterial species Staphylococcus aureus (staph) usually lives harmlessly on human skin, but when introduced to the bloodstream, it can lead to serious infections including pneumonia, toxic shock syndrome, and meningitis. Those infections are very difficult to treat, as staph are quick to adapt to antibiotic chemicals; the multi-antibiotic-resistant infection known as MRSA is a variety of staph.
Trinh’s lab members are researching methods to fight bacterial infections in a more efficient, specific, and adaptable way than applying chemical antibiotics. Their weapon of choice is CRISPR-Cas, a molecular assembly consisting of a guide RNA (gRNA) and a nuclease (DNA-slicing enzyme).


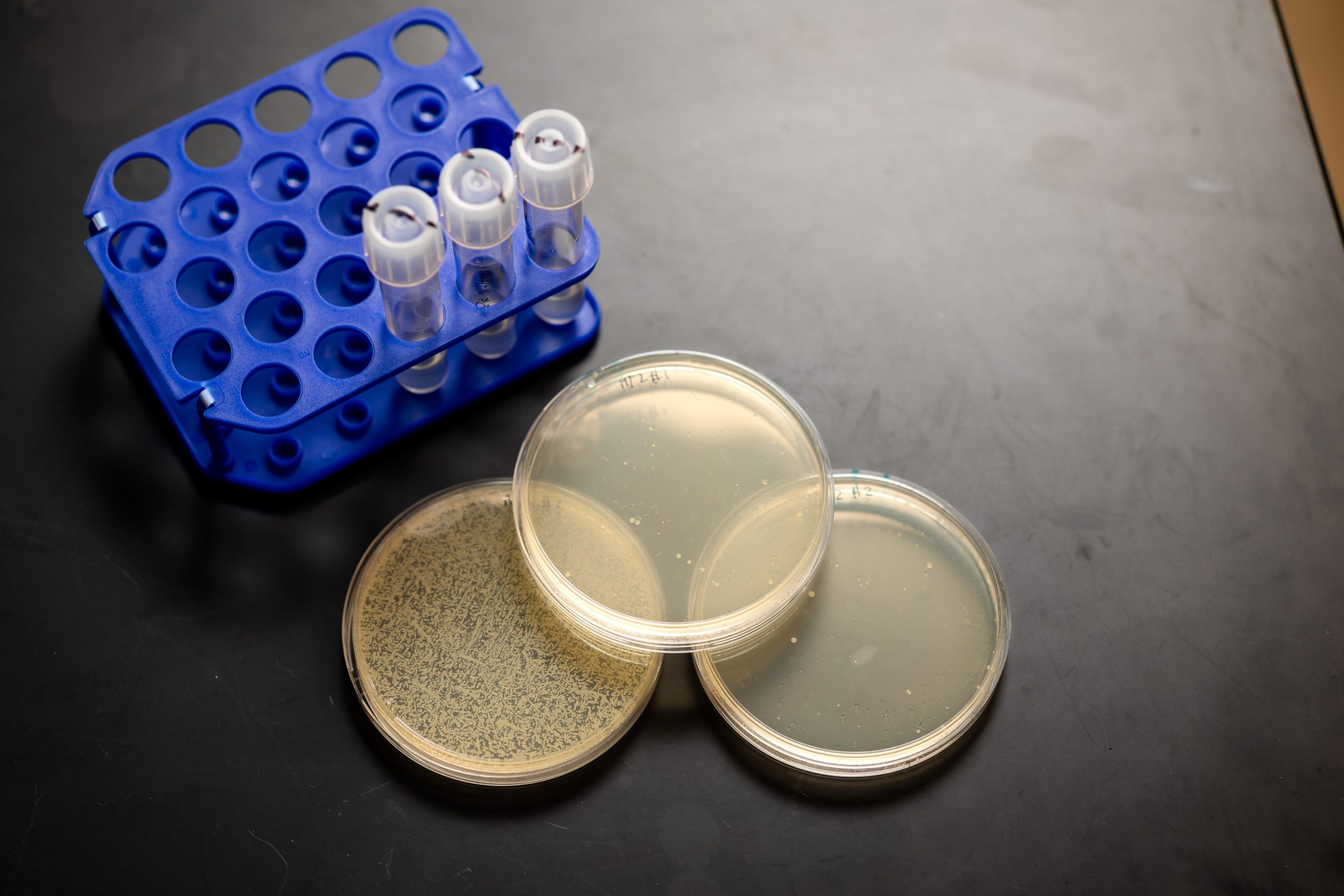
Researchers customize the gRNA to complement a specific sequence on the genome of a target microbe—in this case, staph—and package it into a modified virus that can only attack staph cells. When the gRNA aligns with the matching segment of genomic DNA, the nuclease slices that segment, deactivating it. If that gene is needed for survival, the staph cells have very little opportunity to adapt before they die.
“You don’t want to kill all the bacteria inside of a human,” Boyd said, referring to the billions of beneficial bacteria known to live in the human body. “What’s nice about CRISPR is that since it’s based off of a specific sequence, this kind of targeting isn’t going to affect human cells or bacterial cells in general. It’s only going to affect these specific sequences in staph cells.”
Excited about the technology and its possibilities, Boyd joined Trinh’s lab in early 2024. Working with graduate student David Dooley, she determined to try to make staph-targeting CRISPR even better.
“Instead of just targeting one gene, my CRISPR system uses sequences which are repeated inside the genome, targeting multiple genes at once,” she explained. “There’s a gap in current knowledge about multitargeting gRNAs, so I’m hoping to expand on the existing research.”
Boyd Took Ownership, Earned AURA Grant
To support her summer research, Boyd applied for an Advanced Undergraduate Research Activity (AURA) grant, offered through the UT Division of Student Success. The AURA application includes a two-page summary with goals, methods, and a timeline for the project.
“You kind of have to take ownership for the project,” Boyd said. “I had to make this project more distinct than just ‘helping out.’ That’s why I’m focusing specifically on multi-targeting.”

Receiving the AURA gave Boyd a $2,250 stipend and $250 for research expenses. It also let her dive right into gRNA design.
Using a program called CASPER previously developed by Trinh and postdoctoral researcher Brian Mendoza, Boyd and Dooley identified 30 sequences that are repeated within the staph genome between two and 11 times. Some of these genes encode structures necessary to translate DNA into proteins, while some help staph cells aggregate into colonies.
Once they have a candidate gRNA-CRISPR system assembled, the duo uses it to infect a lab-grown staph population and observe the results. Boyd learned to use Trinh’s cutting-edge Opentron Flex robot to conduct that characterization, which let her screen samples eight times faster than she could manually.
“Hannah is very sharp in learning techniques and is very hard-working,” Trinh said. “By learning this tool, she is already generating quite interesting data for our multitargeting CRISPR project. Her accomplishments this summer have impressed me.”
Biomolecular Engineering Means Problem Solving
Boyd sees a lot of similarities between CRISPR and the medical microbiology research she conducted her freshman year.
“Even though I still need to learn about the computation-based parts of this project, like kinetics analysis, the bio-based part is still a lot of problem solving,” she said. “You’re planning ahead and learning how to juggle many different things at once efficiently and quickly.”
Boyd is grateful that her AURA has given her experience with biological techniques like cutting-edge gene modification in a context that advances medical treatments.
“I feel like I’m actually contributing something to this project that all these other people have spent years on,” she said. “It’s different from doing regular coursework, where it’s just contributing toward my own understanding. I’m helping with something that’s going to be useful in the future. It’s really fulfilling.”
Contact
Izzie Gall (865-974-7203, egall4@utk.edu)
